Utilization of Waste in Organic Synthesis : Novel Synthesis Method Using Phosphine Oxide as Catalyst
1. Purpose
In recent years, our laboratory continues to study the capture of organic molecules based on the formation of hydrogen bonding complexes. In the study of molecular crystals with 2-pyridone derivatives as components, we succeeded in obtaining molecular crystals composed of 2: 2 phenol derivative and dimerized 2-pyridone molecule, and it became clear that they are hydrogen bonded complexes that 2-pyridone molecule adopts the stable arrangement of keto type.
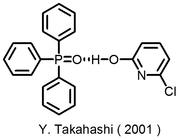
Also, in 2001 Takahashi belonging to our laboratory has found that when a 2-pyridone derivative forms molecular crystals with triphenylphosphine oxide, 2-pyridone molecules adopts the unstable arrangement of enol type via hydrogen bonds between P = O and HO. ( Figure 1 )
This indicates that 2-pyridone occurs in enol form in the absence of a definite acid base. I thought that triphenylphosphine oxide causing this phenomenon could be applied somewhat to the reaction. We came up with the idea that if enolization can occur without using a strong acid or a strong base by using triphenylphosphine oxide, it will be possible to relax the reaction conditions of the organic synthesis reaction including the aldol reaction.
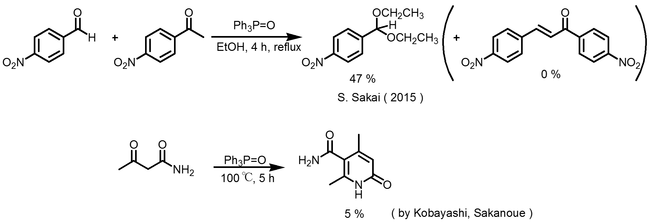
From the past study of our laboratory, it became clear that phosphine oxide, which is adandoned as a byproduct of organic reaction, has the potential to be utilized as a catalyst for the reaction. At the same time I was interested in acetalization and aldol reaction using triphenylphosphine oxide reported by Sakai, Kobayashi and Sakanoue at our laboratory, as well as being attractive to the possibility of its phosphine oxide. ( Scheme 1 )
Also, in 2001 Takahashi belonging to our laboratory has found that when a 2-pyridone derivative forms molecular crystals with triphenylphosphine oxide, 2-pyridone molecules adopts the unstable arrangement of enol type via hydrogen bonds between P = O and HO. ( Figure 1 )
This indicates that 2-pyridone occurs in enol form in the absence of a definite acid base. I thought that triphenylphosphine oxide causing this phenomenon could be applied somewhat to the reaction. We came up with the idea that if enolization can occur without using a strong acid or a strong base by using triphenylphosphine oxide, it will be possible to relax the reaction conditions of the organic synthesis reaction including the aldol reaction.
From the past study of our laboratory, it became clear that phosphine oxide, which is adandoned as a byproduct of organic reaction, has the potential to be utilized as a catalyst for the reaction. At the same time I was interested in acetalization and aldol reaction using triphenylphosphine oxide reported by Sakai, Kobayashi and Sakanoue at our laboratory, as well as being attractive to the possibility of its phosphine oxide. ( Scheme 1 )
2. Describe
Previously Sakai has been studying the reaction time of acetalization at 4 hours, 8 hours, 24 hours and reports that 4 hours is optimal, so whether the same result can be obtained by cyclic acetalization or reaction I examined the time. As a result, it was found that the reaction time of 4 hours obtained with a yield of 33% was optimal.
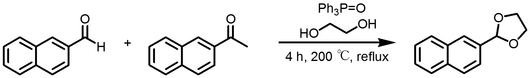
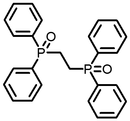
Next, synthesis of cyclic acetal using various diols having OH groups at both ends was investigated. Next I synthesized a cyclic acetal using various diols, naphthalene type aldehydes and ketones which are thought to be easy to react because they are pulling electrons . Even in naphthalene series, a cyclic acetal was similarly synthesized. I studied the synthesis of a compound having two sites of P = O which is a derivative of triphenylphosphine oxide.
Next, synthesis of cyclic acetal using various diols having OH groups at both ends was investigated. Next I synthesized a cyclic acetal using various diols, naphthalene type aldehydes and ketones which are thought to be easy to react because they are pulling electrons . Even in naphthalene series, a cyclic acetal was similarly synthesized. I studied the synthesis of a compound having two sites of P = O which is a derivative of triphenylphosphine oxide.
3. Result
In this experiment, it was revealed that the reaction time is 4 hours even in cyclic acetal synthesis, and even in naphthalene series, acetalization proceeds without acid catalyst in the presence of phosphine oxide. ( Scheme 2 )
As future investigations, I will conduct acetal synthesis using triphenylphosphine oxide as a catalyst, further study of aldol reaction, and examination of reactions using synthesized derivatives. ( Figure 2 )
As future investigations, I will conduct acetal synthesis using triphenylphosphine oxide as a catalyst, further study of aldol reaction, and examination of reactions using synthesized derivatives. ( Figure 2 )